How Are Buffers Important to the Functioning of Living Systems
The weak acid in this buffer is carbonic acid and the salt is sodium bicarbonate. Buffer Systems in Living Organisms.
How Are Buffers Important To The Functioning Of Living Systems Quora
A buffer is a solution that can resist pH change upon the addition of an acidic or basic components.
. Here we examine the basic chemistry of buffer systems and how that chemistry applies to reactions in experimental biological systems. Physiological buffers are chemicals used by the body to prevent large changes in the pH of bodily fluid. To maintain homeostasis of body fluid pH various buffering systems are utilized in addition to proton excretion from the cytosol to the extracellular space and ultimately outside.
Other body organs play important roles in this buffer system. Buffer mixtures are very important in living organisms and the mineral world. It is able to neutralize small amounts of added acid or base thus maintaining the pH of the solution relatively stable.
The pH of a buffer is determined by the Henderson-Hasselbalch equation. It is able to neutralize small amounts of added acid or base thus maintaining the pH of the solution relatively stable. Bicarbonate phosphate hemoglobin and.
However when the basic function of a buffer system resisting changes in hydrogen ion concentration is overlooked experimental artifacts and other problems soon follow. Living organisms need to maintain homeostasis constantly in order to properly grow work and survive. A buffer is a solution that can resist pH change upon the addition of an acidic or basic components.
The Living Fabric 5 The Integumentary System 6 Bones And Skeletal Tissues 7 The Skeleton 8 Joints 9. The lungs get rid of most of the H ions. Many reactions are reversible and will achieve chemical _______ which is the point at which a balance.
Therefore the pH of blood is always maintained at 74. An example of a natural buffer is the blood of mammals. 3 are much greater than concentration of H ions.
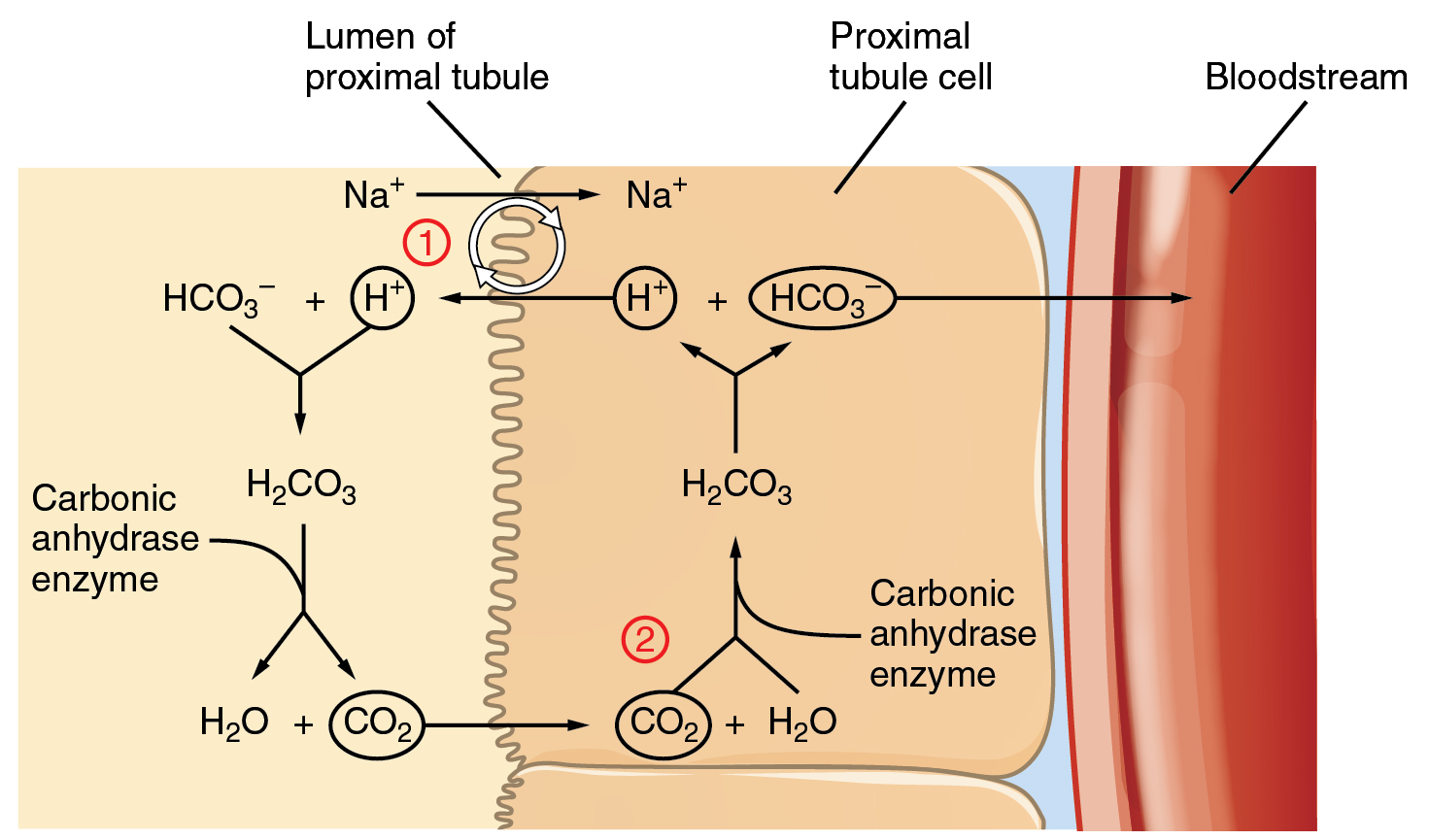
This buffer system involves carbonic acid H 2 CO 3 and bicarbonate HCO 3 anion. Based from the aforementioned examples you may probably already have understood how important homeostasis is. Carbon dioxide is part of a prominent buffer system in the human body.
Role of Buffers in pH Regulation. The Living Units 4 Tissue. It keeps the pH within the proper range.
Buffers are the key. Importance of Buffer System. This buffer works well because concentrations of the buffer components HCO.
Buffers are an important part of the biochemical processes of living things because they help keep the pH within organisms body stable. As mentioned above buffers have a major role in stabilizing the pH of living systems. Describe the buffer action that controls the pH of the cytoplasm and blood.
This is important for processes andor reactions which require specific and stable pH ranges. Buffers readily absorb excess H or OH keeping the pH of the body carefully maintained in the aforementioned narrow range. It always contains free carbonic acid and sodium carbonate.
An Orientation 2 Chemistry Comes Alive 3 Cells. In general homeostasis is essential for normal cell function and overall balance. Regulation of body fluid pH is one of the most important physiological functions of homeostasis because activity of most chemical reactions via enzyme proteins is dependent on fluid pH.
Buffer systems play important roles in nature and in laboratory settings. A substance natural or synthetic that is intended to prevent alleviate or cure an illness is called a n ______. Buffers help keep pH level of solutions at a specific range and it is important for cells to avoid becoming too acidic or too basic based on their metabolism.
This means that changes in the concentration of H ions have little effect on the pH of blood. This is important for processes andor reactions which require specific and stable pH ranges. 1 The Human Body.
In nature they offer protection to living organisms while in labs theyre used to create an environment with a. Every living cell contains natural buffer systems to maintain the constant. The phosphate buffer consists of phosphoric acid H 3 PO 4 in equilibrium with dihydrogen phosphate ion H 2 PO 4 and H.
Buffers are necessary in biological systems because it resist rapid changes in pH when acids and bases are added to it. Vertebrate organisms maintain the pH of blood using a buffer composed of a mixture of carbonic acid H 2 CO 3 and sodium bicarbonate Na HCO 3-. The four Examples of physiological buffers are here.
It plays a very minute role in the blood. The presence of buffers ensures that the bodys pH remains in this range despite changes in the surroundings. Buffers are effective at resisting pH changes when large amounts of acid or base are added to a solution.
Most biochemical reactions that are essential for life only take place in a narrow pH range. 2 formed from H. Because all biological processes are dependent on pH cells and organisms must maintain a specific and constant pH in order to keep their enzymes in the optimum state of protonation.
It is the main buffer in blood plasma and consists of bicarbonate HCO 3 and carbonic acid H 2 CO 3. In labs it is useful to preserve cell compartments from degrading and increases the amount of time they can be reliably used in. The pK for the phosphate buffer is 68 which allows this buffer to function within its optimal buffering range at physiological pH.
Human Anatomy Physiology 11th Edition 11th Edition. The bicarbonate buffer neutralizes stronger dietary and metabolic acids HA converting them into weak bases A with the increase in H 2 CO 3.

Why Are Buffers Important To Living Things Psiberg
How Are Buffers Important To The Functioning Of Living Systems Quora
No comments for "How Are Buffers Important to the Functioning of Living Systems"
Post a Comment